Substrate Ablation of Brugada Syndrome
Indice dell'articolo
Brugada syndrome
Brugada syndrome (BrS) is a genetic disease that mainly affects men in the third and fourth decades of life, and, in the absence of obvious structural heart disease, leads to an increased risk of sudden death due to malignant ventricular arrhythmias (ventricular tachycardia or ventricular fibrillation).
For more details on the disease click here.
Therapeutic options and the rationale for ablative therapy
The main problem in the treatment of this disease is linked to the fact that the cardiac arrest itself can suddenly arise in the absence of warning signs. For this reason, patients discovered to be at the greatest risk for developing fatal arrhythmias can use an implanted defibrillator, which is able to stop a potentially fatal arrhythmia through an electric shock and prevent cardiac arrest.
Although this strategy is extremely effective, it must be admitted that use of these devices comes with the burden of potential side effects, with a significant impact on the quality of life of younger patients.
Furthermore, the defibrillator is a palliative and non-curative therapy of the syndrome, because, although it is extremely effective in interrupting potentially deadly arrhythmias, it is not able to prevent their onset.
Hence, there is the need to identify the mechanisms of the disease itself in order to be able to offer a therapeutic strategy aimed at distorting the natural progression of BrS, to prevent sudden death more effectively.
The discovery of the arrhythmic substrate in BrS
In 2015, for the first time, the group directed by Prof. Pappone demonstrated and identified a group of cells, which express abnormal electrical potentials, on the external surface (epicardial) of the heart at the level of the right ventricle.
These cells are grouped together and form a confluent area, which is found in all patients with BrS.
The characteristics of this abnormal electrical substrate are the basis of the electrical and clinical manifestations of the syndrome itself, explaining the presence of the typical BrS electrocardiogram. In particular, the arrhythmic substrate is associated with the clinical presentation of the disease and the risk of suffering from a more aggressive form of BrS with a more unstable electrical substrate prone to the development of malignant ventricular arrhythmias.
The ablation of the arrhythmic substrate
As previously explained, the BrS arrhythmic substrate is located on the epicardial surface of the right ventricular outflow tract.
To reach this site, it is necessary to perform a subxiphoid puncture (figure 1) and therefore this procedure is performed under general anesthesia.
Figure 1. Epicardial access (video).
Once the pericardial space is reached, a catheter equipped with electrodes with a mapping function that is able to record the electrical activity of the heart is inserted.
Through the use of dedicated software that is associated with mapping systems, it is possible to reconstruct the three-dimensional geometry of the heart and to identify with precision the areas of myocardium affected by the disease (figure 2).
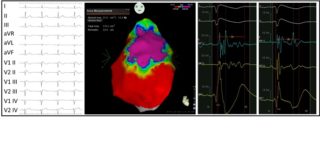
To achieve this goal, it is necessary to administer ajmaline during this mapping phase. This drug is able to unmask the cardiac electrical anomalies to the maximal degree, enabling the visualization and pronounced definition of the area of anomalous substrate to be treated.
Once the anomalous electrical substrate has been identified, it is possible to perform the ablation of this area. Radiofrequency disbursements are rapid and precise, in order to limit ablation to only the outer surface of the heart, enabling the elimination of only surface cells (figure 3).
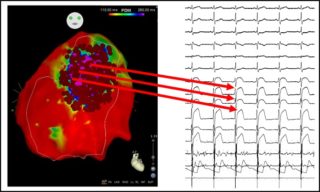
The purpose of the ablation is to eliminate all the anomalous electric potentials located on the epicardium (figure 4), resulting in a complete normalization of the electrocardiogram which no longer shows, after ablation, the classical electrical anomalies of BrS (figure 5). These elements are also associated with the disappearance of malignant ventricular arrhythmias in post-ablation follow-up.
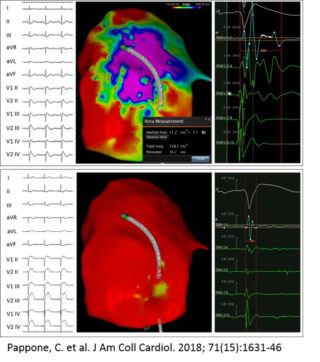
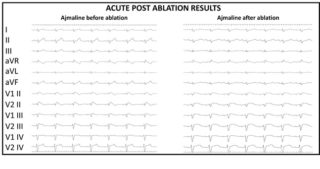
The discovery of the arrhythmic substrate in BrS establishes the first step towards a better knowledge of the disease and towards the development of increasingly effective treatment strategies available to the patient.
If you need information about click here
References
- Pappone C, Brugada J, Vicedomini G, Ciconte G, Manguso F, Saviano M, Vitale R, Cuko A, Giannelli L, Calovic Z, Conti M, Pozzi P, Natalizia A, Crisà S, Borrelli V, Brugada R, Sarquella-Brugada G, Guazzi M, Frigiola A, Menicanti L, Santinelli V. Electrical Substrate Elimination in 135 Consecutive Patients With Brugada Syndrome. Circ Arrhythm Electrophysiol. 2017 May;10(5):e005053. doi: 10.1161/CIRCEP.117.005053. PubMed PMID: 28500178.
- Brugada J, Pappone C, Berruezo A, Vicedomini G, Manguso F, Ciconte G, Giannelli L, Santinelli V. Brugada Syndrome Phenotype Elimination by Epicardial Substrate Ablation. Circ Arrhythm Electrophysiol. 2015 Dec;8(6):1373-81. doi: 10.1161/CIRCEP.115.003220. Epub 2015 Aug 19. PubMed PMID: 26291334.
- Pappone C, Ciconte G, Manguso F, Vicedomini G, Mecarocci V, Conti M, Giannelli L, Pozzi P, Borrelli V, Menicanti L, Calovic Z, Della Ratta G, Brugada J, Santinelli V. Assessing the Malignant Ventricular Arrhythmic Substrate in Patients With Brugada Syndrome. J Am Coll Cardiol. 2018 Apr 17;71(15):1631-1646. doi: 10.1016/j.jacc.2018.02.022. PubMed PMID: 29650119.
- Pappone C, Santinelli V. Brugada Syndrome: Progress in Diagnosis and Management. Arrhythm Electrophysiol Rev. 2019 Mar;8(1):13-18. doi: 10.15420/aer.2018.73.2. PMCID: PMC6434501.
- Pappone C, Brugada J. Ventricular Arrhythmias Ablation in Brugada Syndrome. Current and Future Directions. Rev Esp Cardiol (Engl Ed). 2017 Dec;70(12):1046-1049. doi: 10.1016/j.rec.2017.06.018. PubMed PMID: 28734879.
- Pappone C, Santinelli V. Implantable cardioverter defibrillator and catheter ablation in Brugada syndrome. J Cardiovasc Med (Hagerstown). 2017 Jan;18 Suppl 1:e35-e39. doi: 10.2459/JCM.0000000000000449. PubMed PMID: 27801684.
