Implantable Subcutaneous Defibrillators (S-ICD)
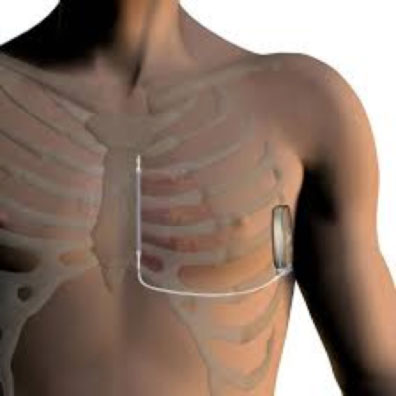
The subcutaneous implantable cardiac defibrillator (S-ICD) is a new type of ICD, produced by the Boston Scientific Company, which does not require the insertion of electrodes inside the heart. The main feature of the S-ICD, compared to classic transvenous ICDs, is that it is exclusively implanted subcutaneously, and effectively performs defibrillation functions, without exposing the risks deriving from the insertion of leads through the venous system up to the cardiac chambers.
Indice dell'articolo
HOW IS THE S-ICD IMPLANTED?
The device is inserted under the skin in the axillary region, while the lead is inserted in the subcutaneous tissue in the sternal region of the chest, anterior to the heart. As with any surgical procedure, the S-ICD implantation is associated with potential risks, such as wound infection and bleeding. Generally, the S-ICD implantation requires a short hospitalization and is performed under local anesthesia, and the intervention lasts about an hour.
WHAT ARE THE ADVANTAGES OF AN S-ICD?
One of the reasons why Subcutaneous ICD was developed is to reduce the risk of complications associated with transvenous leads. Potential complications, such as systemic infections and the need to remove or replace leads in the heart, are in fact minimized or even eliminated with Subcutaneous ICD.
WHAT ARE THE DISADVANTAGES OF AN S-ICD?
S-ICD is not indicated in patients who need cardiac stimulation for bradycardias, tachycardias or cardiac resynchronization. Furthermore, the S-ICD is not suitable in all patients (for example in very young children, or very thin people). Finally, some patients have an ECG morphology that is not correctly recognized by the device, and are therefore not suitable for S-ICD.
| PRO S-ICD | CONTRO S-ICD |
| S-ICD has similar efficacy to transvenous ICDs of stopping ventricular fibrillation (cardiac arrest) | As with any surgical procedure, the S-ICD implantation is associated with potential risks, such as wound infection and bleeding. |
| S-ICD is less invasive, being totally subcutaneous, it leaves the heart and blood vessels intact, and typical complications such as systemic infections and the need to remove or replace leads in the heart are avoided. | S-ICD does not provide anti-bradycardia stimulation: patients with symptomatic bradycardia (sinus node dysfunction or atrioventricular block) require transvenous ICD |
| S-ICD results in less physical limitations: despite the slightly larger case size than a transvenous ICD, it limits the movements of the arm less since it is not implanted under the clavicle. | S-ICD does not provide anti-tachycardia stimulation (ATP): patients with recurrent spontaneous ventricular tachycardia require a traditional transvenous ICD with an anti-tachycardia pacing (ATP) function. |
| S-ICD has a lower risk of breakage compared to the transvenous ICD leads in young subjects who perform relatively intense physical activity | The S-ICD does not provide cardiac resynchronization therapy (CRT). |
| The S-ICD can be implanted using only anatomical reference points, thus eliminating the need for fluoroscopy (and consequently exposure to radiation). | Some patients have an ECG morphology that is not correctly recognized by the device, and therefore are not suitable for S-ICD |
| Some people may experience discomfort when sleeping on the left side | |
| In women, S-ICD could create discomfort in wearing a bra | |
| In very thin people, the S-ICD could create discomfort because the case could be prominent or very visible. |
FOR WHICH PATIENTS IS AN S-ICD DEVICE INDICATED?
The patients who benefit most from the implantation of a subcutaneous defibrillator (S-ICD) are young patients in the absence of structural heart disease. In fact, in these patients, who would be exposed to the risks involved with having an implanted device for a prolonged period of time because of their young age, having a subcutaneous defibrillator without electrodes inside the heart makes their future less complicated. Furthermore, the need for anti-tachycardia or anti-bradycardia stimulation is less likely in these patients.
CAN A PATIENT ASK TO RECEIVE AN S-ICD INSTEAD OF A TRADITIONAL ICD?
Unless an ICD is implanted in an emergency condition, a patient can express the preference for an S-ICD instead of a traditional transvenous ICD. However, the recommendation and the final choice on the type of ICD to be implanted (subcutaneous vs. transvenous) is the responsibility of the electrophysiologist, who will take into account the patient’s preferences, but also all the clinical characteristics of the patient and the disease.
IS REMOTE MONITORING OF AN S-ICD POSSIBLE?
Remote monitoring of the S-ICD can be performed through the Latitude system, with specific methods that will be explained by the Remote Monitoring Service.
If you want to know more:
