Anticoagulant drugs
Anticoagulant drugs are drugs used to prevent thrombus formation and to hinder the growth of blood clots that have already formed. Thrombus formation can lead to the occlusion of the vessel or, in the case where the thrombus detaches from the vessel wall and enters the bloodstream, giving rise to a thromboembolic phenomenon, it can lead to very serious and sometimes fatal consequences, including myocardial and pulmonary infarction and cerebral stroke.
Indice dell'articolo
HOW ANTICOAGULANT DRUGS WORK
Unlike platelet anti-aggregating agents that act on platelet aggregation, anticoagulant drugs interfere with the coagulation process (the so-called coagulation cascade) by acting on co-factors and coagulation factors whose activation leads to the formation of a fibrin network that traps blood cells, giving rise to the clot.
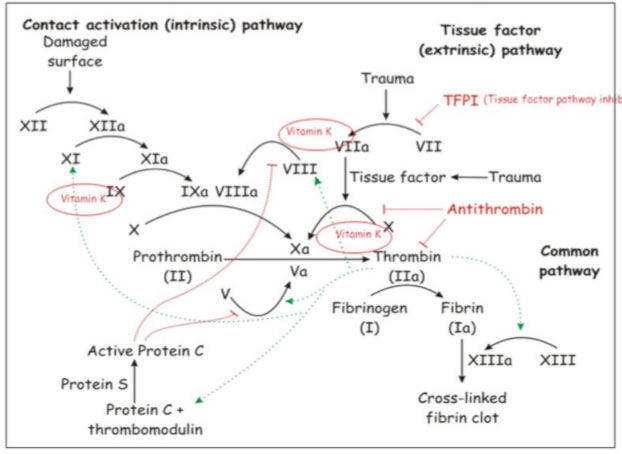
Currently there are different types of anticoagulant drugs, administered orally or parenterally, which can be divided according to the mechanism of action and according to their chemical structure, in the following way:
- Heparin parenteral anticoagulants: are activators of antithrombin III, a physiological inhibitor of coagulation, including heparin and its derivatives (called low molecular weight heparins, LMWH), administered exclusively parenterally.
- Oral coumarin-type anticoagulants: are vitamin K antagonists (vitamin implicated in the function of various factors of the coagulation cascade, AVK), e.g. Warfarin (Coumadin) or Acenocumarol (Sintrom), administered orally
- Direct acting oral anticoagulants (DAOC): are direct antagonists of coagulation factors:
- factor IIa inhibitors: are antagonists of factor IIa (thrombin), preventing the formation of the fibrin network that traps erythrocytes and gives rise to the blood clot, for example Dabigatran (Pradaxa), administered orally
- Anticoagulants factor Xa inhibitors: are antagonists of factor Xa, hindering the transformation of prothrombin I into thrombin (factor IIa), for example Apixaban (Eliquis), Rivaroxaban (Xarelto) and Endoxaban (Lixiana) administered orally
WHAT ARE THE INDICATIONS FOR ORAL ANTICOAGULANT MEDICINES?
Oral anticoagulants, according to the current guidelines, are prescribed in various clinical situations, both acute and chronic:
- in primary and secondary prevention and in the treatment of venous thromboembolism and pulmonary embolism;
- in the acute phase of peripheral arterial occlusions (acute coronary syndrome, unstable angina, myocardial infarction, cerebral stroke)
- in the prevention of arterial thromboembolism in patients with non-valvular atrial fibrillation (AF) and in patients undergoing electrical cardioversion;
- in the primary and secondary prevention of stroke in the context of atrial fibrillation
- in patients with mechanical heart valve prostheses (to prevent thrombus formation on the valves);
HOW EPARINIC DRUGS WORK AND WHEN TO USE THEM
Heparin is an endogenous compound, naturally present within organisms, located in the granules of mast cells, plasma and other tissues, consisting of a heterogeneous mixture of sulfated mucopolysaccharides (sulphated glycosaminoglycans).
The heparinic preparations carry out their anticoagulant action by binding to antithrombin III, a physiological inhibitor of the coagulation process, enhancing the inhibitory action against coagulation factors Xa and IIa (thrombin). When heparin binds to antithrombin III, it causes an increase in the affinity and binding speed of antithrombin III itself with factors Xa and IIa. The anticoagulant action physiologically exerted by antithrombin III, therefore, appears to be considerably increased by the administration of heparin drugs.
Heparin (of animal origin) has been made available as a medicine. In particular, lower molecular weight derivatives have been obtained, which are administered exclusively parenterally by injection or infusion, depending on the case.
The heparin anticoagulant drugs currently used are basically two types:
- Standard or unfractionated heparin (high molecular weight), is characterized by a rapid onset of the anticoagulant effect but for a short duration of action. For this reason, it is also used in emergencies and requires infusion therapy.
- Low molecular weight heparins (LMWH or LMWH, from Low Molecular Weight Heparin), have a longer duration of action than standard heparin, and are therefore used in chronic use and provide one or two daily administrations, generally subcutaneously. Examples of the most used active ingredients are:
- Enoxaparina (Clexane®, Clexane T®);
- Nadroparina (Fraxodi®, Fraxiparina®, Seledie®, Seleparina®);
Given the need for parenteral administration, heparin is generally used in the acute phase and not in the chronic prevention of thromboembolism. The therapeutic applications of heparin are therefore in the prevention and acute management of arterial and venous thromboembolism in the following conditions:
- atrial fibrillation (in conjunction with electrical or pharmacological cardioversion, during ablation, etc)
- deep vein thrombosis (for the prevention and management of pulmonary thromboembolism, TEP)
- acute arterial obstruction (in particular acute coronary syndrome and ischemic stroke)
- major surgery (especially orthopedics)
- hemodialysis.
HOW ANTICUMARINIC DRUGS WORK AND WHEN TO USE THEM
The most commonly prescribed drugs in chronic oral anticoagulant (TAO) therapy are still vitamin K antagonists (AVK). In particular, warfarin was the gold standard for decades in the prevention and treatment of these diseases.
Cumarinic anticoagulant drugs interfere with the redox cycle of vitamin K, a very important cofactor in the synthesis and activation of some vitamin K-dependent coagulation factors, such as factor II (prothrombin) and factors VII, IX and X The oxidation-reduction cycle of vitamin K involved in the blood coagulation process involves the intervention of different enzymes: vitamin K quinone reductase and vitamin K 2,3-epoxide reductase.
Cumarinic anticoagulant drugs are so defined because, from the chemical point of view, they are derivatives of coumarin (a natural organic compound), and act as inhibitors of vitamin K 2,3-epoxide reductase. The AVK drugs currently used in Italy are:
- Warfarin (Coumadin®);
- Acenocumarolo (Sintrom®).
Given that the dose of anticoagulants required is highly variable among different individuals, it is necessary to regularly measure the coagulation time through a blood test, the INR (International Normalized Ratio), to be sure that the treatment is effective, safe, and the right dosage. The higher the value of the INR, the greater the fluidity of the blood.
The normal physiological value of the INR in a healthy subject is between 0.8 and 1.2 (the value may differ slightly from one laboratory to the other).
Depending on the pathology, different therapeutic ranges are prescribed. For example, in a patient in TAO for atrial fibrillation, the value of INR should be between 2 and 3. In patients with heart valves, an INR value between 3 and 4 is instead recommended.
Many drugs and foods can change the bioavailability of AVK drugs, and therefore make the value of INR highly variable. Therefore, patients in TAO must regularly undergo blood tests to check the value of the INR, since this must be adjusted if necessary so that the blood does not become too liquid or too thick.
The doctor who follows the patient then sets up a treatment plan according to an individual schedule (weekly / monthly) and sets the appropriate therapeutic amount to be taken daily (eg 1/2 tablet, or 1/4, etc.)
The side effects that can occur during therapy with coumarin anticoagulants are many, particularly:
- increased risk of bleeding and the appearance of bleeding complications in different organs and tissues;
- allergic reactions in sensitive individuals;
- nausea and vomiting, abdominal pain;
- ecchymoses and hematomas;
- alopecia (hair loss);
- liver and kidney damage
The problems of using AVK drugs have led, in recent years, to replacing them with the new oral anticoagulant drugs (NAO). However there are at least two categories of patients in which the use of TAO is still recommended:
- patients with mechanical heart valve prostheses
- patients with severe chronic renal failure (GFR glomerular filtrate between 29 and 15 ml / min)
HOW THE NEW DIRECT ORAL ANTICOAGULANT DRUGS (DOAC) WORK AND WHEN TO USE THEM
The new oral anticoagulants (NAO), also called anticoagulants direct inhibitors (DOAC), include two types of drugs:
- direct inhibitors of factor IIa or thrombin (dabigatran), which exert their anticoagulant effect by binding directly to the active site of the thrombin, thus inhibiting the downstream effects;
- direct factor Xa inhibitors (endoxaban, apixaban and rivaroxaban), which inhibit factor Xa, simultaneously blocking the intrinsic pathway and the extrinsic pathway of the coagulation “cascade”
From the analysis of the studies comparing the efficacy and safety of the new oral anticoagulant drugs versus the coumarin drugs (warfarin) in the treatment of non-valvular atrial fibrillation (FANV), the following advantages have emerged:
- 50% reduction in the risk of hemorrhagic stroke
- incidence of stroke and systemic embolism reduced by 18% without any increase in the risk of major bleeding
- reduced incidence of intracranial bleeding and increased incidence of gastrointestinal bleeding
Furthermore, NAOs have been shown to be safe and effective compared to traditional therapies in other cardiological fields, as in the case of dabigatran, used in antithrombotic therapy following angioplasty with stent insertion and in non-valvular atrial fibrillation subjected to electrical cardioversion.
The use of the new oral anticoagulants does not provide for a routine monitoring of coagulation due to the predictable nature of their pharmacokinetics. The dosage and the dosage interval, according to the guidelines, must not be changed in relation to the coagulation parameters.
The only tests to be performed are related to kidney function once a year or every six months, if: the patient is over 80 years old, is at high risk of bleeding, has moderate renal insufficiency or is in conditions that may impair kidney function.
Marco Moja proposed an interesting review on DOAC https://www.slideshare.net/anemo_site/1330-moia-140307-2).
NAOs have a bioavailability that varies depending on the drug used: dabigatran 37%, apixaban 50%, endoxaban 62%, rivaroxaban 66% fasted and 100% on a full stomach. The onset of the anticoagulant effect is rapid (a few hours compared to 4-5 days with warfarin) and the half-life is short: it varies from 5 to 17 hours.
NAOs are associated with lower drug and dietary interactions than warfarin. The interactions found are with the P-glycoprotein transporter (P-gp) and with the cytochrome P450 isoenzyme 3A4 (CYP3A4). Currently, no scientific evidence is available regarding the possible risks in the co-administration of food or herbal medicines and NAOs (Di Minno et al., 2017).
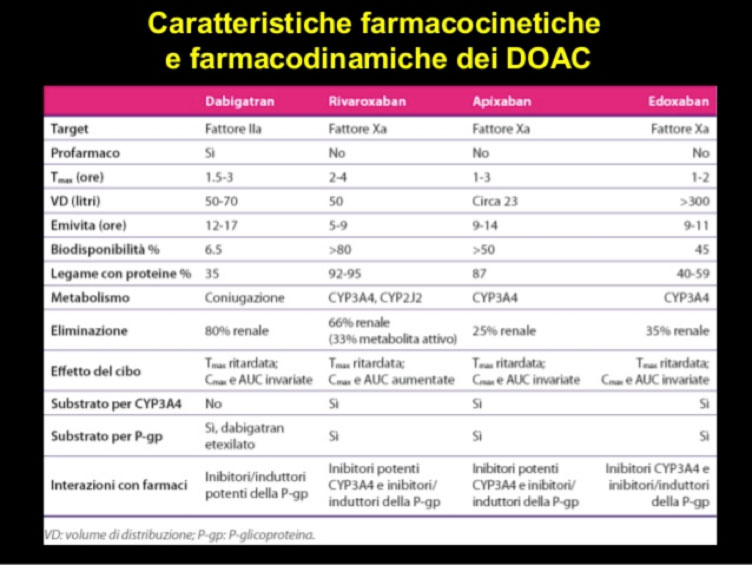
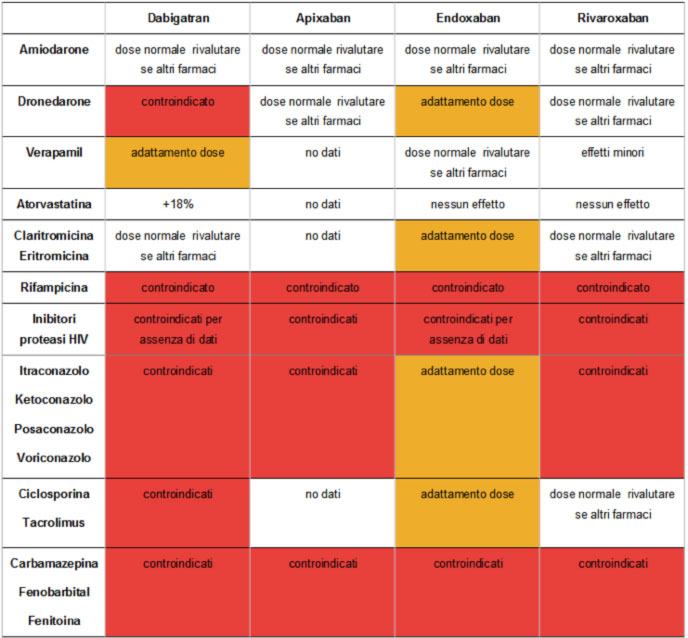
The main drug interactions of NAOs are with some anticonvulsants / antiepileptics, antifungals, antibiotics and HIV protease inhibitors:
Emergencies The blood concentration of DOACs and the related anticoagulant activity can be measured in emergency situations (severe bleeding, thrombosis, need for surgery or in specific clinical conditions) by specific tests depending on the type of anticoagulant used. The analyzes that can be performed for direct thrombin inhibitors are: aPTT; the thrombin time (TT), which is very sensitive to the presence of dabigatran; ecarin coagulation time (ECT); the diluted thrombin time (dTT). The specific laboratory tests indicated for factor Xa inhibitors are: prothrombin time (PT), which provides quantitative information regarding rivaroxaban, and chromogenic anti-FXa.
The positive aspect of DOAC in the event of hemorrhage is the short half-life. If there is a need to quickly counteract the anticoagulant effect of these drugs, you can do so by using:
- non-specific agents: the prothrombin complex concentrate (PCC), the activated PCC and perform hemodialysis in the event of an overdose of dabigatran;
- specific agents: the only one currently approved is idarucizumab, a monoclonal antibody that is able to bind dabigatran and neutralize its anticoagulant effect. Alpha andexanet and aripazine, both potential antidotes of factor Xa inhibitors, are under development and approval.
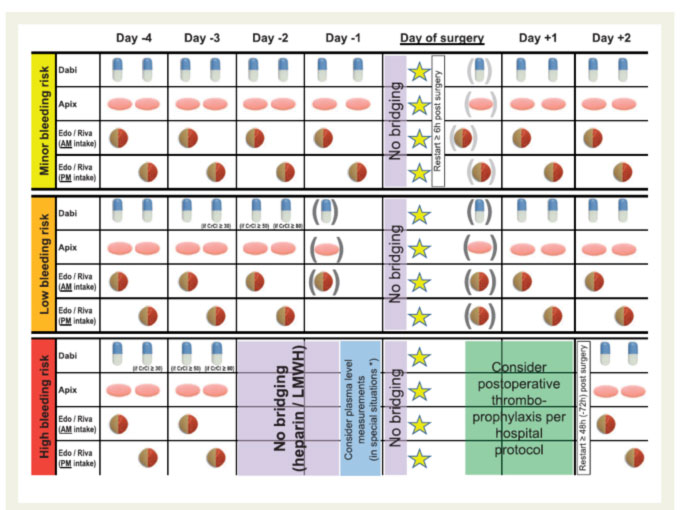
Perioperative management
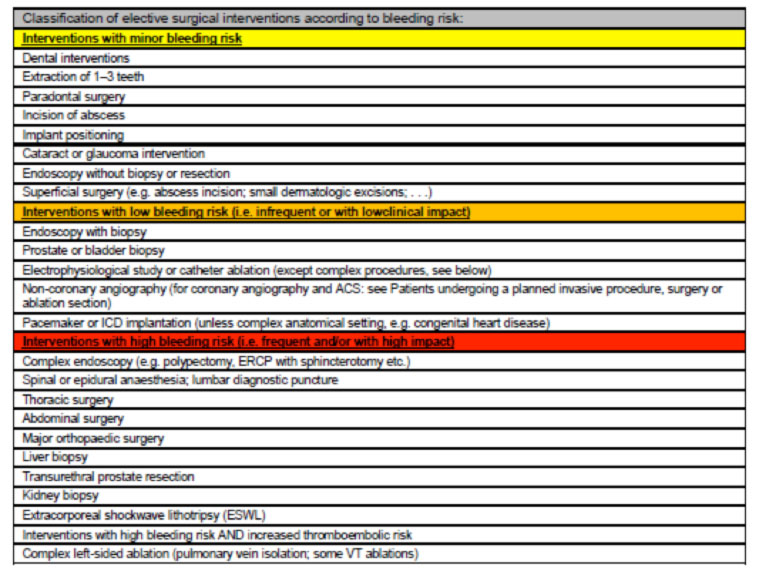
The management of DOAC before and after a surgical operation varies according to the bleeding risk of the procedure and to the patient’s renal function:
- in procedures without any risk of clinically relevant bleeding and / or for which it is possible to perform an effective local haemostasis (e.g. dental procedures) are carried out 12 or 24 hours after the last intake, depending on the dosage interval, for then resume therapy after 6-8 hours.
- in procedures with a clinically relevant risk of bleeding:
- If the patient has normal renal function, the last dose is indicated 24 hours before surgery if the risk of bleeding is low, while 48 hours before if the risk is high;
- for patients with creatinine clearance between 15-30 mL / min it is recommended to consider interruption 36 hours earlier for low risk bleeding procedures and 48 hours earlier if the risk is high;
- in invasive procedures requiring a certain period of entrapment, heparin is administered at low molecular weight after 6-8 hours, if adequate haemostasis has taken place, and then the therapy with DOAC is restarted after 48-72 hours
Side effects
Possible side effects related to oral anticoagulant therapy include gastrointestinal bleeding, intraocular hemorrhage and alopecia. However, these effects are also associated with traditional anticoagulants, and are generally reversible with drug withdrawal.
Advantages of DOACs
DOACs facilitate adherence to therapy, thanks to a reduction in the limits imposed by the practical management of therapy, such as the absence of routine monitoring of blood tests, the fixed dosage regimen, the need for specialized centers nearby, and the minor dietary and pharmacological interactions.
However, the fact that there is no test capable of providing information regarding the overall adherence to therapy (such as the INR for vitamin K antagonists), in addition to the short half-life and the absence of frequent controls, makes it crucial to prevent over- and under-dosing.
Therapeutic plan
The prescription of DOAC drugs is carried out after the preparation of the treatment plan carried out on the AIFA site by the reference Cardiologist. The treatment plan has a duration of 26 or 52 weeks. For the repetition of the treatment plan, it is necessary to carry out a follow-up visit with the specialist cardiologist of reference, bringing the following recent examinations:
- Blood exam
- Renal function (creatininemia and GFR)
- Liver function (Alanine transaminase (ALT or SGPT) and Aspartate transaminase (AST or SGOT)
| Recommendations to patients on DOAC therapy
For the prevention of episodes of overdose and underdose, which could lead to the development of thromboembolic or hemorrhagic complications, patients receiving DOAC are recommended to:
|
BIBLIOGRAPHY
– Auyeung V., Patel J.P., Abdou J.K., Vadher B., Bonner L., Brown A., Roberts L.N., Patel R.K. and Arya R. (2016), Anticoagulated patient’s perception of their illness, their beliefs about the anticoagulant therapy prescribed and the relationship with adherence: impact of novel oral anticoagulant therapy – study protocol for The Switching Study: a prospective cohort study, BMC Hematology, 16(1): 9.
– Camm J., Knight E., Izzat L.M. and Hospital P.P. (2012), The Route Map for Change & the European Atlas on the Prevention of AF-Related Stroke, 1-250.
– Cannon C.P., Bhatt D.L., Oldgren J., Lip G.Y.H., Ellis S.G., Kimura T., Maeng M., Merkely B., Zeymer U., Gropper S., Nordaby M., Kleine E., Harper R., Manassie J., Januzzi J.L., ten Berg J.M., Steg P. G., Hohnloser S.H. (2017), Dual Antithrombotic Therapy with Dabigatran after PCI in Atrial Fibrillation, New England Journal of Medicine, 1–12.
– Di Minno A., Frigerio B., Spadarella G., Ravani A., Sansaro D., Amato M., Kitzmiller J.P., Pepi M., Tremoli E., Baldassarre D. (2017), Old and new oral anticoagulants: Food, herbal medicines and drug interactions, Blood Reviews, 31(4):193–203.
– Dogliotti A., Paolasso E., Giugliano R.P. (2013), Novel oral anticoagulants in atrial fibrillation: A meta-analysis of large, randomized, controlled trials vs warfarin, Clinical Cardiology, 36(2): 61–67.
– Eikelboom J.W., Connolly S.J., Bosch J., Dagenais G.R., Hart R.G., Shestakovska O., Diaz R., Alings M., Lonn E.M., Anand S.S., Widimsky P., Hori M., Avezum A., Piegas L.S., Branch K.R.H., Probstfield J., Bhatt D.L., Zhu J., Liang Y., Maggioni A.P., Lopez-Jaramillo P., O’Donnell M., Kakkar A., Fox K.A.A., Parkhomenko A.N., Ertl G., Störk S., Keltai M., Ryden L., Pogosova N., Dans A.L., Lanas F., Commerford P.J., TorpPedersen C., Guzik T.J., Verhamme P.B., Vinereanu D., Kim J.H., Tonkin A.M., Lewis B.S., Felix C., Yusoff K., Steg P.G., Metsarinne K.P., Cook Bruns N., Misselwitz F., Chen E., Leong D. and Yusuf S. (2017), Rivaroxaban with or without Aspirin in Stable Cardiovascular Disease, New England Journal of Medicine, 1-12.
– Ezekowitz M.D., Pollack C.V., Sanders P., Halperin J.L., Spahr J., Cater N., Petkun, W., Breazna A., Kirchhof P. and Oldgren J. (2016), Apixaban compared with parenteral heparin and/or vitamin K antagonist in patients with nonvalvular atrial fibrillation undergoing cardioversion: Rationale and design of the EMANATE trial, American Heart Journal, 179: 59–68.
– Heidbuchel H., Verhamme P., Alings M., Antz M., Diener H.C., Hacke W., Oldgren ,J., Sinnaeve P., Camm A.J., Kirchhof P. (2015) Updated European Heart Rhythm Association Practical Guide on the use of non-vitamin K antagonist anticoagulants in patients with non-valvular atrial fibrillation. Europace, 17(10): 1467-507.
